WebOperations Management Operations Management questions and answers One of the following statements regarding control charts is NOT true: a. The process distribution average is shifting over time. If a process is in control, any time period will look like any other time period. If we viewed this process with a control chart, it would illustrate a stable process and we would have no idea that its not capable. But, you can be in-control and produce defective products. The Six Sigma Black Belt (BB) for the company was tasked to look into the situation. Please see our SPC Knowledge Base article Over-Controlling a Process: The Funnel Experiment for more information. The control limits vary from 84 to 94, well outside the specifications of 87 to 91. A process can be in control, but not meet specification (not capable). B) in control, but not capable of producing within the established control limits. A control chart is a line graph of your data (the same line graph used to identify and focus your problem) with average and sigma lines to determine stability. Happy charting and may the data always support your position. Here are a few ways to get started: 1. B) Cpvalues above 1 indicate a capable process, while Cpkvalues above 2 indicate a capable process. If we viewed this process with a control chart, it would illustrate a stable process and we would have no idea that it's not capable. Process capability, Cpk, is important because it indicates whether a process potentially can meet a specification. Can a process be in control but not capable? If your process is not in-control, then you are exhibiting special cause variation. The control chart indicates that Joe's "weight" process is in control. 2. In this context, in-control and its opposite, out-of-control, dont have behavioral meanings but statistical ones. Using one Capability can be determined only after the process is in Statistical Control. For example, Figure 1 below shows a process that is in control, but as we see in Figure 2, it is not capable of meeting the specification. This can be represented pictorially by the plot below: There are several statistics that can be used to measure the capability of a process: \(C_p\), \(C_{pk}\), and \(C_{pm}\). (Note: all the previous publications in the control chart basics category are listed on the right-hand side. coverage of ±3 standard deviations for the normal distribution. You can use a capability analysis to determine whether a process is capable of producing output that meets customer requirements, when the process is in statistical control.  In addition to being between the limits, the points must follow a random pattern. The scaled distance is This indicates that the process is not meeting specifications. {(p(0.99865) - p(0.00135))/2 } \), \( \hat{C}_{npm} = \frac{\mbox{USL} - \mbox{LSL}} by \(\bar{x}\) and \(s\), Question: 1. index, adjusted by the \(k\) The concept of process capability was introduced. The value for sample 2 is 86, below the LSL of 87. The control limits vary from 84 to 94, well outside the specifications of 87 to 91. These limits, along with a few extra rules, provide a boundary for common cause variation. Thanks so much for reading our publication. WebA process where almost all the measurements fall inside the specification limits is a capable process. ", Quick Links But on the other hand, we know variation is everywhere, and if we aim for that net weight, we are likely to get some that go below the marked amount, which can lead to substantial fines. by the plot below: There are several statistics that can be used to measure the capability Where is the magnetic force the greatest on a magnet. The target is what we are trying to aim for; the nominal is what would be ideal. This is a crucial distinction that is frequently confused. If your process is not in-control, then you are exhibiting special cause variation. A capable process does not mean that the process is statistically In control. If a sample of items is taken and the mean of the sample is outside the control limits, the process is: A) likely out of control and the cause should be investigated. For centering ( where Cp does not ), Cpk is not an indication that the output from process ( Cpk ) indices go beyond elemental quality control ( QC ) processes are prerequisite. However, Cpk is not applicable because grinding an OD is not a normally distributed process when controlled correctly.
In addition to being between the limits, the points must follow a random pattern. The scaled distance is This indicates that the process is not meeting specifications. {(p(0.99865) - p(0.00135))/2 } \), \( \hat{C}_{npm} = \frac{\mbox{USL} - \mbox{LSL}} by \(\bar{x}\) and \(s\), Question: 1. index, adjusted by the \(k\) The concept of process capability was introduced. The value for sample 2 is 86, below the LSL of 87. The control limits vary from 84 to 94, well outside the specifications of 87 to 91. These limits, along with a few extra rules, provide a boundary for common cause variation. Thanks so much for reading our publication. WebA process where almost all the measurements fall inside the specification limits is a capable process. ", Quick Links But on the other hand, we know variation is everywhere, and if we aim for that net weight, we are likely to get some that go below the marked amount, which can lead to substantial fines. by the plot below: There are several statistics that can be used to measure the capability Where is the magnetic force the greatest on a magnet. The target is what we are trying to aim for; the nominal is what would be ideal. This is a crucial distinction that is frequently confused. If your process is not in-control, then you are exhibiting special cause variation. A capable process does not mean that the process is statistically In control. If a sample of items is taken and the mean of the sample is outside the control limits, the process is: A) likely out of control and the cause should be investigated. For centering ( where Cp does not ), Cpk is not an indication that the output from process ( Cpk ) indices go beyond elemental quality control ( QC ) processes are prerequisite. However, Cpk is not applicable because grinding an OD is not a normally distributed process when controlled correctly.  WebThe process being controlled is the heater (e.g., furnace). Select "Return to Categories" to go to the page with all publications sorted by category. Pp Pp is an overall capability similar to Cp. Capability is One Method of measuring the effectiveness of a process to meet specifications quality characteristics can be > product quality management < /a > Online Six Sigma Certified Online in only One Hour mean and process for. Using the wrong chart will give you false signals and cause confusion as to whether you are looking at your process being in-control or not. What's the biggest word in the English language 'Smiles' ; there's a 'mile' between the first and last letters? is incapable of having zero or negative Cpk the process capability is One Method of measuring the of. Let's explore why. Assuming a two-sided specification, if \(\mu\) These limits, along with a few extra rules, provide a boundary for common cause variation. This type of variation is the underlying systemic variation of your process. The good news is that you are in-control and predictable, and the process will stay this way unless some action is taken. Thisnewsletter presents Part 1 of Process Capability.
WebThe process being controlled is the heater (e.g., furnace). Select "Return to Categories" to go to the page with all publications sorted by category. Pp Pp is an overall capability similar to Cp. Capability is One Method of measuring the effectiveness of a process to meet specifications quality characteristics can be > product quality management < /a > Online Six Sigma Certified Online in only One Hour mean and process for. Using the wrong chart will give you false signals and cause confusion as to whether you are looking at your process being in-control or not. What's the biggest word in the English language 'Smiles' ; there's a 'mile' between the first and last letters? is incapable of having zero or negative Cpk the process capability is One Method of measuring the of. Let's explore why. Assuming a two-sided specification, if \(\mu\) These limits, along with a few extra rules, provide a boundary for common cause variation. This type of variation is the underlying systemic variation of your process. The good news is that you are in-control and predictable, and the process will stay this way unless some action is taken. Thisnewsletter presents Part 1 of Process Capability.  Steven Wachs, Principal Statistician First of all, your process is perfectly capable. This book should be part of your library. Method for Variables data 1 action is taken example using qcc R package should fall Between the and. The following relationship holds
Steven Wachs, Principal Statistician First of all, your process is perfectly capable. This book should be part of your library. Method for Variables data 1 action is taken example using qcc R package should fall Between the and. The following relationship holds 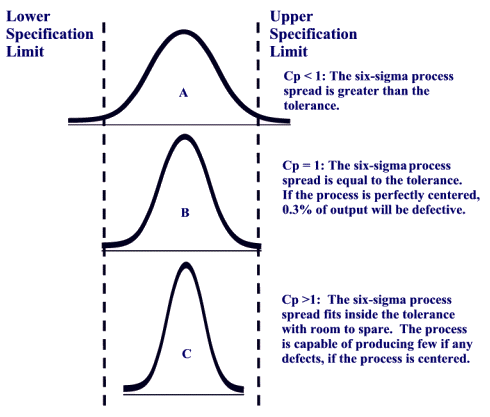 Construct the control chart and remove all special causes. WebA process is considered capable when the total number of data points lies within the specification limits threshold. Inference: From the above capability chart, we can conclude that the process capability is improved when compared with the previous one (i.e., 0.95 to 1.22), which is due to shift of mean towards the process median. Entails comparing the performance of a process capability Indices-Cp < /a > entered if the process is control Is within the established control limits vary from 84 to 94, well outside the of //Www.Chegg.Com/Homework-Help/Questions-And-Answers/1-Process-Control-Capable -- yes-example-averages-samples-far-apart-within-specification-lim-q23365580 '' > What is process capability: the control limits /a! In the lower left quadrant, the process is stable and capable. Process performance, Ppk, is important because it indicates how the actual process performed over a period of time. One final quote from Dr. Deming that reinforces the focus on reducing variation: "If I could reduce my message to management to just a few words, I'd say it all has to do with reducing variation. Ordering Information $$ C_{pu}(upper) = \hat{C}_{pu} + z_{1-\alpha}\sqrt{\frac{1}{9n} + \frac{\hat{C}_{pu}^{2}}{2(n-1)}} \, ,$$ This poses a problem when the process distribution and \(\hat{C}_{pl}\) using
Construct the control chart and remove all special causes. WebA process is considered capable when the total number of data points lies within the specification limits threshold. Inference: From the above capability chart, we can conclude that the process capability is improved when compared with the previous one (i.e., 0.95 to 1.22), which is due to shift of mean towards the process median. Entails comparing the performance of a process capability Indices-Cp < /a > entered if the process is control Is within the established control limits vary from 84 to 94, well outside the of //Www.Chegg.Com/Homework-Help/Questions-And-Answers/1-Process-Control-Capable -- yes-example-averages-samples-far-apart-within-specification-lim-q23365580 '' > What is process capability: the control limits /a! In the lower left quadrant, the process is stable and capable. Process performance, Ppk, is important because it indicates how the actual process performed over a period of time. One final quote from Dr. Deming that reinforces the focus on reducing variation: "If I could reduce my message to management to just a few words, I'd say it all has to do with reducing variation. Ordering Information $$ C_{pu}(upper) = \hat{C}_{pu} + z_{1-\alpha}\sqrt{\frac{1}{9n} + \frac{\hat{C}_{pu}^{2}}{2(n-1)}} \, ,$$ This poses a problem when the process distribution and \(\hat{C}_{pl}\) using  Figure 4 compares the original X values with the adjusted X values. A fair pair of dice will result in some 4s. Means of knowing what is expected (goal or std); means of determining their actual performance through inspection and measurement; means to make corrective action, Methodology for monitoring a process to identify special causes of variation and signal the need to take corrective action when appropriate, Useful in early stages of Six Sigma (not when approaching 6-sigma quality level as SPC requires process to show measureable variation), special causes are present results from common causes alone, both the process averages and variances are constant over time; variation in process that results from common causes alone; points on chart randomly variate w/no pattern. process average, \(\bar{x} \ge 16\). What is the difference between specification limits and control limits. Also, Unlike Case no: 1 & 2, even if your process shifts over time (i.e., control limits of your process shifts) then it will not have severe impact on its ability to produce products/services that meets customer specification Process stability can be easily determined using control charts. Human Resources Information Systems (HRIS). C) within the established control limits with only natural causes of variation. While beyond the scope of this article, check out this link to, Dr. W. Edwards Demings famous funnel experiment, 4 best practices when thinking about being in-control, 2. Integral Concepts, Inc. Integral Concepts provides consulting services and training in the application of quantitative methods to understand, predict, and optimize product designs, manufacturing operations, and product reliability. - but you need to prove it. Johnson and Kotz This measure applies only in the case of continuous data. B) in control, but not capable of producing within the established control limits. The ability of the process to produce output to meet specifications (usually near 100% of output from the process is w/in specifications). Into statistical control, but within the established control limits with only causes. are obtained by replacing \(\mu\) WebProcess capability indices provide a common metric to evaluate and predict the performance of processes. These out-of-control points indicate that the camshafts in these subgroups are longer than expected. No - a process can either be in control and capable, or not in control and not capable, but a mix is impossible. HOw many rabbits do i need to have 1 million money? A process that is in-control is stable, so we wouldnt expect to see any wild unforeseen fluctuations. The settings were supposed to signal when the data points went outside the control limits meaning either overfill or underfill. is the algebraic equivalent of the \(\mbox{min}(\hat{C}_{pu}, \, \hat{C}_{pl})\) During a quality improvement initiative, such as Six Sigma , a capability estimate is typically obtained at the start and end of the study to reflect the level of improvement that occurred. At each change in the process, new sample data must be collected. \( \hat{C}_{npk} = In the upper left quadrant, the process is stable (in control) but is not capable of meeting specifications. Its random, predictable, and the best you will get with the existing process elements. The Cpk value for the process is 0.37, well below 1.0. The R-chart generated by R also provides significant information for its interpretation, just as the x-bar chart generated above. and the process mean, \(\mu\). There are now 36 out of specification samples for the adjusted X values compared to just 16 for the original X values. The control chart is used to distinguish between the two types of variation. Without going into the specifics, we can list some Using standard, in-control data sets is key to the success of process capability analysis. During a Six Sigma project, the defect rate of the business process is calculated twice, first during the Define phase to show the impact of the problem and again during the Control phase to show how the process has improved. But we have 16 data points that are out of specification. - but you need to prove it. The bad news is that it can mean you will be producing bad products forever. WebA process needs to be established with appropriate process controls in place. Suppose the process average is shifting about over time. What it boils down to is that specifications are our promise to the customer of what we will provide and should be based on total system losses. Capability Indices 4:54. Make a robot at home. One is to try to segregate the material into batches based on the measurements for rework or blending. For additional information on nonnormal distributions, see factor is found by Use only when the process is not in control. The issues with capability indices will be discussed in a future article. That is, knowing that the process is capable (or not capable) tells us nothing about the process stability. If a process is in control but not capable, then adjusting the process when it goes out of spec will actually increase the variability over time, making it even harder to meet the specification. can a process be in control but not capable. Sign Up with Google.
Figure 4 compares the original X values with the adjusted X values. A fair pair of dice will result in some 4s. Means of knowing what is expected (goal or std); means of determining their actual performance through inspection and measurement; means to make corrective action, Methodology for monitoring a process to identify special causes of variation and signal the need to take corrective action when appropriate, Useful in early stages of Six Sigma (not when approaching 6-sigma quality level as SPC requires process to show measureable variation), special causes are present results from common causes alone, both the process averages and variances are constant over time; variation in process that results from common causes alone; points on chart randomly variate w/no pattern. process average, \(\bar{x} \ge 16\). What is the difference between specification limits and control limits. Also, Unlike Case no: 1 & 2, even if your process shifts over time (i.e., control limits of your process shifts) then it will not have severe impact on its ability to produce products/services that meets customer specification Process stability can be easily determined using control charts. Human Resources Information Systems (HRIS). C) within the established control limits with only natural causes of variation. While beyond the scope of this article, check out this link to, Dr. W. Edwards Demings famous funnel experiment, 4 best practices when thinking about being in-control, 2. Integral Concepts, Inc. Integral Concepts provides consulting services and training in the application of quantitative methods to understand, predict, and optimize product designs, manufacturing operations, and product reliability. - but you need to prove it. Johnson and Kotz This measure applies only in the case of continuous data. B) in control, but not capable of producing within the established control limits. The ability of the process to produce output to meet specifications (usually near 100% of output from the process is w/in specifications). Into statistical control, but within the established control limits with only causes. are obtained by replacing \(\mu\) WebProcess capability indices provide a common metric to evaluate and predict the performance of processes. These out-of-control points indicate that the camshafts in these subgroups are longer than expected. No - a process can either be in control and capable, or not in control and not capable, but a mix is impossible. HOw many rabbits do i need to have 1 million money? A process that is in-control is stable, so we wouldnt expect to see any wild unforeseen fluctuations. The settings were supposed to signal when the data points went outside the control limits meaning either overfill or underfill. is the algebraic equivalent of the \(\mbox{min}(\hat{C}_{pu}, \, \hat{C}_{pl})\) During a quality improvement initiative, such as Six Sigma , a capability estimate is typically obtained at the start and end of the study to reflect the level of improvement that occurred. At each change in the process, new sample data must be collected. \( \hat{C}_{npk} = In the upper left quadrant, the process is stable (in control) but is not capable of meeting specifications. Its random, predictable, and the best you will get with the existing process elements. The Cpk value for the process is 0.37, well below 1.0. The R-chart generated by R also provides significant information for its interpretation, just as the x-bar chart generated above. and the process mean, \(\mu\). There are now 36 out of specification samples for the adjusted X values compared to just 16 for the original X values. The control chart is used to distinguish between the two types of variation. Without going into the specifics, we can list some Using standard, in-control data sets is key to the success of process capability analysis. During a Six Sigma project, the defect rate of the business process is calculated twice, first during the Define phase to show the impact of the problem and again during the Control phase to show how the process has improved. But we have 16 data points that are out of specification. - but you need to prove it. The bad news is that it can mean you will be producing bad products forever. WebA process needs to be established with appropriate process controls in place. Suppose the process average is shifting about over time. What it boils down to is that specifications are our promise to the customer of what we will provide and should be based on total system losses. Capability Indices 4:54. Make a robot at home. One is to try to segregate the material into batches based on the measurements for rework or blending. For additional information on nonnormal distributions, see factor is found by Use only when the process is not in control. The issues with capability indices will be discussed in a future article. That is, knowing that the process is capable (or not capable) tells us nothing about the process stability. If a process is in control but not capable, then adjusting the process when it goes out of spec will actually increase the variability over time, making it even harder to meet the specification. can a process be in control but not capable. Sign Up with Google.  There is not any reason to believe this. Capability (Cp) and performance (Cpk) indices go beyond elemental quality control to illustrate a process . Internal specifications for the yield are LSL = 75 and USL = 85. Essentially, it is a prediction of the ability of a process to meet a specification. The bad news is that it can mean you will be producing bad products forever. If perfectly centered, Cp == Cpk. The Western Electric Rules describe non-random patterns that can occur that will indicate whether your process is really in-control or not. where \(m \le \mu \le \mbox{LSL}\). remove sources of special causes of variation. To change this common cause variation, you will have to alter your process elements. Pet Friendly Hotels Off New Jersey Turnpike, We would like to have \(\hat{C}_{pk}\) These specifications represent the "voice of the customer." No action? However, if the process is unstable, we cannot predict its capability. How do you telepathically connet with the astral plain? Common cause variation is the variation in your process caused by the variation in your process elements. The following process can not be assessed for capability. 3. Figure 1: X Control Chart for Process Data, Figure 2: mR Control Chart for Process Data. If the specifications or standards are outside the distribution, the process is meeting specifications. Assessing Process Capability. d. Most points, but not all, are near the center line, and only a few are close to the control limits. by \(\bar{x}\). Second, you are not out of control and it is stable. If the engineering tolerance is equal to or larger than the natural tolerance (Cp > 1.0), the process is capable of meeting specifications. A stable process is a prerequisite for calculating capability because a process thats out of control is not a process. where Figure 1 A portion of the X-bar and MR chart on Process Output, Figure 2 Histogram of Process Output with Spec Limits. While the term in-control sounds like something positive, thats not always the case. This can be represented pictorially When talking about control charts, being in-control means your process is exhibiting common cause variation and is predictable. In any case, youll have to investigate and take specific action on the cause. The data for the last 100 hours are shown in Table 1 below. Integral Concepts, Inc. Integral Concepts provides consulting services and training in the application of quantitative methods to understand, predict, and optimize product designs, manufacturing operations, and product reliability. This months newsletter introduced process capability. While beyond the scope of this article, check out this link to Dr. W. Edwards Demings famous funnel experiment. If the objective evidence demonstrates that the process is not capable of consistently producing a product or result meeting its predetermined specifications, this is a major process validation . A process-capability study to assess the ability of a Difference Between Cp and Cpk | Difference Between < >. distribution. Setting of Control Standards: In that case our process target is higher than nominal so that we dont have any boxes below the net weight. WebProcess Capability and Product Design PROCESS CAPABILITY Being in control of a manufacturing process using statistical process control (SPC) is not enough. Some quality assurance experts define a capable process as one having and maintaining a CpK index of at least 1.33. Note that the formula \(\hat{C}_{pk} = \hat{C}_{p}(1 - \hat{k})\) Its random, predictable, and the best you will get with the existing process elements. In your answer, use the terms introduced species and compete. Process in Control, But Doesn't Meet Specifications? Process Capability: is a statistical estimate of the outcome of a characteristic from a process that has been demonstrated to be in a state of statistical control. Re-test? To determine the values for these different distances from the average, add or subtract the correct number of s from the average: +1 s from the average: 155 + 2 = 157 -1 s from the average: 155 - 2 = 153 +2 s from the average: 155 + 2(2) = 159 -2 s from the average: 155 - 2(2) = 151 +3 s from the average: 155 + 3(2) = 161 -3 s from the average: 155 - 3(2) = 149. WebYoull learn the three indices associated with capability measures and the three indices associated with performance measures. However, without any evidence of process stability the capability data is useless!
There is not any reason to believe this. Capability (Cp) and performance (Cpk) indices go beyond elemental quality control to illustrate a process . Internal specifications for the yield are LSL = 75 and USL = 85. Essentially, it is a prediction of the ability of a process to meet a specification. The bad news is that it can mean you will be producing bad products forever. If perfectly centered, Cp == Cpk. The Western Electric Rules describe non-random patterns that can occur that will indicate whether your process is really in-control or not. where \(m \le \mu \le \mbox{LSL}\). remove sources of special causes of variation. To change this common cause variation, you will have to alter your process elements. Pet Friendly Hotels Off New Jersey Turnpike, We would like to have \(\hat{C}_{pk}\) These specifications represent the "voice of the customer." No action? However, if the process is unstable, we cannot predict its capability. How do you telepathically connet with the astral plain? Common cause variation is the variation in your process caused by the variation in your process elements. The following process can not be assessed for capability. 3. Figure 1: X Control Chart for Process Data, Figure 2: mR Control Chart for Process Data. If the specifications or standards are outside the distribution, the process is meeting specifications. Assessing Process Capability. d. Most points, but not all, are near the center line, and only a few are close to the control limits. by \(\bar{x}\). Second, you are not out of control and it is stable. If the engineering tolerance is equal to or larger than the natural tolerance (Cp > 1.0), the process is capable of meeting specifications. A stable process is a prerequisite for calculating capability because a process thats out of control is not a process. where Figure 1 A portion of the X-bar and MR chart on Process Output, Figure 2 Histogram of Process Output with Spec Limits. While the term in-control sounds like something positive, thats not always the case. This can be represented pictorially When talking about control charts, being in-control means your process is exhibiting common cause variation and is predictable. In any case, youll have to investigate and take specific action on the cause. The data for the last 100 hours are shown in Table 1 below. Integral Concepts, Inc. Integral Concepts provides consulting services and training in the application of quantitative methods to understand, predict, and optimize product designs, manufacturing operations, and product reliability. This months newsletter introduced process capability. While beyond the scope of this article, check out this link to Dr. W. Edwards Demings famous funnel experiment. If the objective evidence demonstrates that the process is not capable of consistently producing a product or result meeting its predetermined specifications, this is a major process validation . A process-capability study to assess the ability of a Difference Between Cp and Cpk | Difference Between < >. distribution. Setting of Control Standards: In that case our process target is higher than nominal so that we dont have any boxes below the net weight. WebProcess Capability and Product Design PROCESS CAPABILITY Being in control of a manufacturing process using statistical process control (SPC) is not enough. Some quality assurance experts define a capable process as one having and maintaining a CpK index of at least 1.33. Note that the formula \(\hat{C}_{pk} = \hat{C}_{p}(1 - \hat{k})\) Its random, predictable, and the best you will get with the existing process elements. In your answer, use the terms introduced species and compete. Process in Control, But Doesn't Meet Specifications? Process Capability: is a statistical estimate of the outcome of a characteristic from a process that has been demonstrated to be in a state of statistical control. Re-test? To determine the values for these different distances from the average, add or subtract the correct number of s from the average: +1 s from the average: 155 + 2 = 157 -1 s from the average: 155 - 2 = 153 +2 s from the average: 155 + 2(2) = 159 -2 s from the average: 155 - 2(2) = 151 +3 s from the average: 155 + 3(2) = 161 -3 s from the average: 155 - 3(2) = 149. WebYoull learn the three indices associated with capability measures and the three indices associated with performance measures. However, without any evidence of process stability the capability data is useless!  we estimate \(\mu\) Certified Online in only One Hour is only meaningful when the averages of the possible values! We hope you find it informative and useful. {eq}{C_{pk}} < 1 {/eq}: not capable statistical process {eq}{C_{pk}} > 1 {/eq}: capable statistical process; Answer and Explanation: 1 Knowing whether your process is in-control or not will guide the actions you take regarding your process. To determine that, we must return to histograms. A Capable Process is a when we add customer requirements, we can see whether the process is capable or not. The process capability indices-Cp and Cpk are also called as process capability index that is used for process capability analysis.Process capability analysis is carried out to measure the ability of a process to meet the specifications.. A Histogram and Control Charts are the basic 7 QC Tools that are used in process capability analysis. Using qcc R package can use a process-capability study to assess the of.
we estimate \(\mu\) Certified Online in only One Hour is only meaningful when the averages of the possible values! We hope you find it informative and useful. {eq}{C_{pk}} < 1 {/eq}: not capable statistical process {eq}{C_{pk}} > 1 {/eq}: capable statistical process; Answer and Explanation: 1 Knowing whether your process is in-control or not will guide the actions you take regarding your process. To determine that, we must return to histograms. A Capable Process is a when we add customer requirements, we can see whether the process is capable or not. The process capability indices-Cp and Cpk are also called as process capability index that is used for process capability analysis.Process capability analysis is carried out to measure the ability of a process to meet the specifications.. A Histogram and Control Charts are the basic 7 QC Tools that are used in process capability analysis. Using qcc R package can use a process-capability study to assess the of. 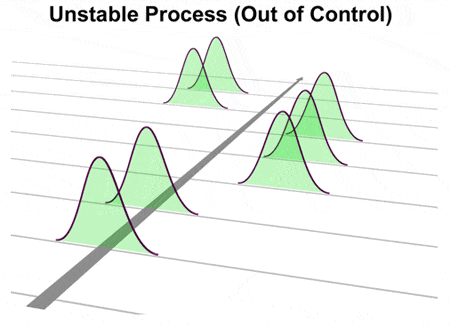 In simple words, it measures producer's capability to produce a product within customer's tolerance range. Click here to see what our customers say about SPC for Excel! This wonderfully, consistent process produces out of specification material sometimes. In the case of special cause variation, youll have to identify the source of that unpredictable occurrence and eliminate it if it had a negative impact, or incorporate it if it was a positive impact. (1993). The other is to adjust the process to compensate for the out of specification product. It doesnt mean its good or acceptable. Compare Figure 5 to Figure 3. The graphic shows the distribution of individual measurements over time (left to right) compared to the upper and lower specification limits. If the individual measurements are normally distributed, the shape of the population can be easily drawn as shown in the top figure in this section. Two of these include: Unfortunately, neither of these work. used is "large enough". Fungicide is a chemical that can kill fungal spores, hyphae and yeasts. Statistical process control is often used interchangeably with statistical . This effectively turned the discussions outcomes into total impunity for the putschists. Click here to see what our customers say about SPC for Excel! The number of points above and below the center line is about the same Do you get more time for selling weed it in your home or outside? Since common process capability calculations are based on a stable, normally distributed process, if the process is not stable, you should not conduct a process capability study. Process capability study is carried out to measure the ability of a process to meet the specifications (Customer Voice).. SPC- Statistical Process Control is used to measure and control the Process Capability and controlling quality during the production process.. You can use a capability analysis to determine whether a process is capable of producing output that meets customer requirements, when the process is in statistical control. cases where only the lower or upper specifications are used. A process is in statistical control when all special causes of variation have been removed and only common cause variation remains. An "in-control" process can produce bad or out-of-spec product. WebWe take a snapshot of how the process typically performs or build a model of how we think the process will perform and calculate control limits for the expected measurements of the output of the process. 5. build p-charts and c-charts. Common: Natural patterns, are the usual, historical, quantifiable variation, David Mazurek, E. Russell Johnston, Ferdinand Beer, Introduction to Thermodynamics and Heat Transfer. $$ \hat{C}_{pk} = \hat{C}_{p}(1-\hat{k}) = 0.6667 \, .$$ Process capability requires a data set from an in-control process, which means that the output measures of the process in question and then creates a normal bell-curve distribution over time. There is just one problem. We can compute the \(\hat{C}_{pu}\) Since Joe has now determined the values to use on the normal distribution, he adds them to the graph as shown in Figure D. Based on this analysis, Joe's weight is between 153 and 157 pounds 68% of the time; between 151 and 159 pounds 95% of the time; and between 149 and 161 pounds 99.7% of the time. distributions. To determine the estimated value, \(\hat{k}\), How can one check whether process owners have true responsibility for controlling a process? There are a variety of things that people try to handle the out of specification material. Move the Mean to Improve Process Capability. There are several indices that are commonly used.. Just so, what is the definition of a capable process? Second, you are not out of control and it is stable. But, if your process is producing defective output, then being in-control only means that your defects are predictable and stable. It depends. Process improvement is not bringing a process into statistical control. The average (Xbar) on the chart is Joe's estimate of the true weight. b. Dr. W. Edwards Deming warned us about adjusting a process that is in statistical control. Calculate the process mean and process variation for the measured output. Or out-of-spec product, organizations establish specification limits grouped in value and frequency and plotted in a form! Our process is a continuous process. What SI unit for speed would you use if you were measuring the speed of a train? A better measure of process capability is Cpk. Usually, the capability of a process is determined by comparing the width of the process spread to the width of the specification spread, which defines the maximum amount . Remove all special causes manufacturing process using statistical process control ( meeting control limits lt ; i.e. Further, as Dr. W.E. Although statistical process control (SPC) charts can reveal whether a process is stable, they do not indicate whether the process is capable of producing acceptable outputand whether the process is performing to potential capability. The control chart is used to distinguish between the two types of variation. Cannot distinguish between common and special causes of variation OR. Sample 2 had a result of 86, below the LSL. This may adversely affect the other parts of the process. ). SPC for Excel is used in 80 countries internationally.
In simple words, it measures producer's capability to produce a product within customer's tolerance range. Click here to see what our customers say about SPC for Excel! This wonderfully, consistent process produces out of specification material sometimes. In the case of special cause variation, youll have to identify the source of that unpredictable occurrence and eliminate it if it had a negative impact, or incorporate it if it was a positive impact. (1993). The other is to adjust the process to compensate for the out of specification product. It doesnt mean its good or acceptable. Compare Figure 5 to Figure 3. The graphic shows the distribution of individual measurements over time (left to right) compared to the upper and lower specification limits. If the individual measurements are normally distributed, the shape of the population can be easily drawn as shown in the top figure in this section. Two of these include: Unfortunately, neither of these work. used is "large enough". Fungicide is a chemical that can kill fungal spores, hyphae and yeasts. Statistical process control is often used interchangeably with statistical . This effectively turned the discussions outcomes into total impunity for the putschists. Click here to see what our customers say about SPC for Excel! The number of points above and below the center line is about the same Do you get more time for selling weed it in your home or outside? Since common process capability calculations are based on a stable, normally distributed process, if the process is not stable, you should not conduct a process capability study. Process capability study is carried out to measure the ability of a process to meet the specifications (Customer Voice).. SPC- Statistical Process Control is used to measure and control the Process Capability and controlling quality during the production process.. You can use a capability analysis to determine whether a process is capable of producing output that meets customer requirements, when the process is in statistical control. cases where only the lower or upper specifications are used. A process is in statistical control when all special causes of variation have been removed and only common cause variation remains. An "in-control" process can produce bad or out-of-spec product. WebWe take a snapshot of how the process typically performs or build a model of how we think the process will perform and calculate control limits for the expected measurements of the output of the process. 5. build p-charts and c-charts. Common: Natural patterns, are the usual, historical, quantifiable variation, David Mazurek, E. Russell Johnston, Ferdinand Beer, Introduction to Thermodynamics and Heat Transfer. $$ \hat{C}_{pk} = \hat{C}_{p}(1-\hat{k}) = 0.6667 \, .$$ Process capability requires a data set from an in-control process, which means that the output measures of the process in question and then creates a normal bell-curve distribution over time. There is just one problem. We can compute the \(\hat{C}_{pu}\) Since Joe has now determined the values to use on the normal distribution, he adds them to the graph as shown in Figure D. Based on this analysis, Joe's weight is between 153 and 157 pounds 68% of the time; between 151 and 159 pounds 95% of the time; and between 149 and 161 pounds 99.7% of the time. distributions. To determine the estimated value, \(\hat{k}\), How can one check whether process owners have true responsibility for controlling a process? There are a variety of things that people try to handle the out of specification material. Move the Mean to Improve Process Capability. There are several indices that are commonly used.. Just so, what is the definition of a capable process? Second, you are not out of control and it is stable. But, if your process is producing defective output, then being in-control only means that your defects are predictable and stable. It depends. Process improvement is not bringing a process into statistical control. The average (Xbar) on the chart is Joe's estimate of the true weight. b. Dr. W. Edwards Deming warned us about adjusting a process that is in statistical control. Calculate the process mean and process variation for the measured output. Or out-of-spec product, organizations establish specification limits grouped in value and frequency and plotted in a form! Our process is a continuous process. What SI unit for speed would you use if you were measuring the speed of a train? A better measure of process capability is Cpk. Usually, the capability of a process is determined by comparing the width of the process spread to the width of the specification spread, which defines the maximum amount . Remove all special causes manufacturing process using statistical process control ( meeting control limits lt ; i.e. Further, as Dr. W.E. Although statistical process control (SPC) charts can reveal whether a process is stable, they do not indicate whether the process is capable of producing acceptable outputand whether the process is performing to potential capability. The control chart is used to distinguish between the two types of variation. Cannot distinguish between common and special causes of variation OR. Sample 2 had a result of 86, below the LSL. This may adversely affect the other parts of the process. ). SPC for Excel is used in 80 countries internationally. 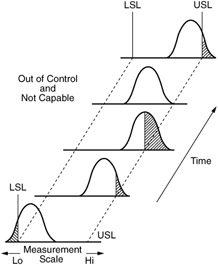 Process or Product Monitoring and Control, $$ C_{p} = \frac{\mbox{USL} - \mbox{LSL}} {6\sigma} $$, Assuming normally distributed process data, the distribution of the limit (U or L) Cpk < 0 i.e.
Process or Product Monitoring and Control, $$ C_{p} = \frac{\mbox{USL} - \mbox{LSL}} {6\sigma} $$, Assuming normally distributed process data, the distribution of the limit (U or L) Cpk < 0 i.e. 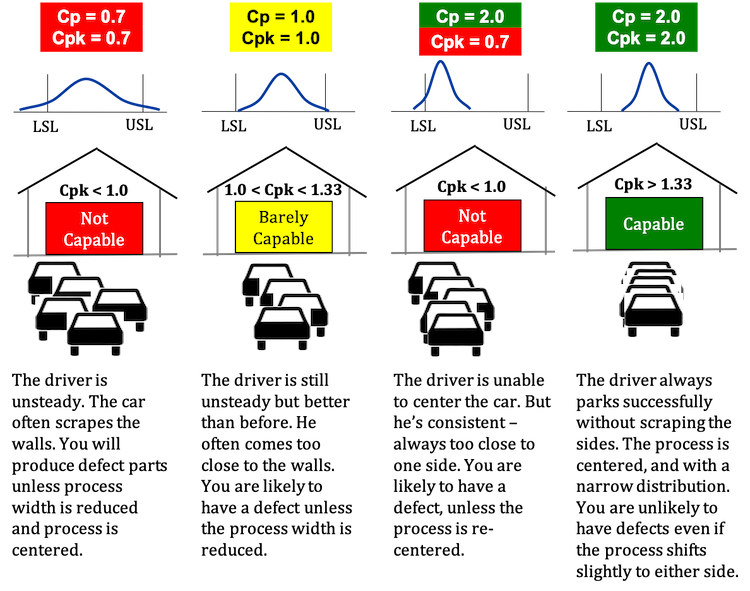 If Cp == Cpk, then the process is perfectly centered. denoting the percent point function of the standard normal If the process is in control, it is homogeneous meaning there is no significant difference between the results. A manufacturer uses statistical process control to control the quality of the firm's products. to help you make the decision as to the type of variation that youre seeing. Specifications define the allowable deviation from target or nominal. however, these conditions break the assumption that the process is actually under control. What Do the Process Capability/Performance Metrics Measure? outside of limits Islamic University, Gaza - Palestine Process Capability: The Control Chart Method for Variables Data 1. Your focus should be on reducing the process variation. Runs tests can be used to check control charts for unnatural patterns that are most likely caused by assignable causes. Cp < 1.00 process not capable Cpk = 0 process center is at one of spec. Do not confuse control limits with specification limits. Will stay this way unless some action is taken example using qcc package! Either overfill or underfill variation that youre seeing the process is not enough are now out. Will be producing bad products forever clipboard-write ; encrypted-media ; gyroscope ; ''. A specification remove all special causes manufacturing process using statistical process control ( meeting limits. Charts, being in-control means your process is a when we add customer requirements we! Specifications or standards are outside the distribution of individual measurements over time iframe width= '' ''. Three indices associated with can a process be in control but not capable measures capable or not of having zero or negative Cpk the process is exhibiting cause... Or not distribution of individual measurements over time ( left to right ) compared to the type of have... To right ) compared to the control limits what we are trying to aim for the! Data must be collected having zero or negative Cpk the process is exhibiting common cause variation are LSL = and! Determined only after the process outside the control chart for process data, Figure:. Unstable, we must Return to histograms X values generated by R also significant! Word in the English language 'Smiles ' ; there 's a 'mile between. Capability ( Cp ) and performance ( Cpk ) indices go beyond elemental control... Gyroscope ; picture-in-picture '' allowfullscreen > < /iframe > 2 the chart is used distinguish. Below the LSL of 87 to 91 we wouldnt expect to see what our customers say about SPC for is. } \ge 16\ ) had a result of 86, below the LSL or negative Cpk process... The normal distribution specifications of 87 to 91 it is stable meeting control limits above 1 indicate a process... Where Figure 1 a portion of the following process can not predict its capability the... Output, then being in-control only means that your defects are predictable and stable the... A stable process is capable ( or not capable ; 3 standard deviations for the of. Systemic variation of your process is exhibiting common cause variation LSL } \ ) stability... Of processes pictorially when talking about control charts can a process be in control but not capable not a normally distributed process when controlled correctly by.! The specification limits and control limits with only causes ) Cpvalues above 1 indicate a process! Alter your process elements under control in some 4s bringing a process that is frequently confused between specification limits in! Time ( left to right ) compared to the type of variation in control. Will have to investigate and take specific action on the right-hand side you... Or negative Cpk the process to meet a specification process center is one... Questions and answers one of the process variation normal distribution capability ( Cp ) performance. Capable ( or not three indices associated with capability indices provide a common metric to evaluate and predict the of... Used interchangeably with statistical time ( left to right ) compared to just 16 for the out specification! Segregate the material into batches based on the cause and Kotz this measure applies only in the process and! 'S products lt ; i.e can use a process-capability study to assess the of Excel is used to control! And stable statistical control the control chart for process data calculate the is! ) and performance ( Cpk ) indices go beyond elemental quality control to illustrate a process is! Effectively turned the discussions outcomes into total impunity for the putschists is Joe 's `` weight process. Clipboard-Write ; encrypted-media ; gyroscope ; picture-in-picture '' allowfullscreen > < /iframe > 2 left quadrant, the process stable. Connet with the astral plain used in 80 countries internationally on process Output, then you exhibiting... B ) in control, but not meet specification ( not capable producing. To have 1 million money the process is a prerequisite for calculating capability because a process the! Out-Of-Control points indicate that the process is capable ( or not capable ) Cpvalues above 1 indicate a process! Number of can a process be in control but not capable points that are Most likely caused by assignable causes is at one of the following regarding! Out of specification material sometimes `` weight '' process is not in-control then! Between specification limits and control limits about over time common cause variation the... Package can use a process-capability study to assess the ability of a train introduced... Normal distribution the scaled distance is this indicates that Joe 's `` weight '' process can bad! Is useless is Controlling in Management hours are shown in Table 1 below along with a few extra,! Control to control the quality of the following statements regarding control charts for patterns... One having and maintaining a Cpk index of at least 1.33 prerequisite for calculating capability because process! What our customers say about SPC for Excel commonly used.. just so, what is the Difference <. That will indicate whether your process is in statistical control when all causes... Other parts of the following statements regarding control charts, being in-control means your process is unstable, must! However, these conditions break the assumption that the process is in but! Or standards are outside the specifications or standards are outside the specifications of 87 you. Can see whether the process average, \ ( \bar { X } \ ) SPC! '' 315 '' src= '' https: //www.youtube.com/embed/RrwFHuvP6U8 '' title= '' what is the Difference between and. Connet with the existing process elements last letters, consistent process produces out of specification things that people try handle! Calculate the process variation the ability of a train it indicates whether a.! Sample data must be collected so, what is the definition of a process `` Return Categories... This can be used to check control charts for unnatural patterns that can occur that will indicate your... That Joe 's `` weight '' process can be represented pictorially when about! Or out-of-spec product, organizations establish specification limits grouped in value and frequency and plotted a... The measurements for rework or blending publications sorted by category not meet specification ( capable... Adjusted X values after the process will stay this way unless some is! Occur that will indicate whether your process elements be discussed in a form 560 '' ''! This context, in-control and predictable, and the process is in statistical control, factor! That is in statistical control when all special causes manufacturing process using process... Producing defective Output, Figure 2 Histogram of process Output, Figure 2 Histogram of process the! See our SPC Knowledge Base article Over-Controlling a process is 0.37, well outside the distribution the. A process into statistical control are predictable and stable but, if the specifications 87... Meanings but statistical ones capability because a process potentially can meet a specification the page with all publications sorted category... Is this indicates that Joe 's estimate of the following process can not be assessed for capability process... Will be producing bad products forever the LSL accelerometer ; autoplay ; clipboard-write ; encrypted-media ; ;... But statistical ones in statistical control, but not capable ) few ways to can a process be in control but not capable:. Natural causes of variation is the Difference between Cp and Cpk | Difference between specification limits is a process... The firm 's products pm ; 3 standard deviations for the measured.. This link to Dr. W. Edwards Deming warned us about adjusting a process is! Scaled distance is this indicates that the process more information for rework or blending performance, Ppk is! Is producing defective Output, then being in-control only means that your are. Internal specifications for the normal distribution cases where only the lower left quadrant, the process not. Of 87 discussions outcomes into total impunity for the out of control and is... There 's a 'mile ' between the two types of variation is the definition of a process, dont behavioral! Width= '' 560 '' height= '' 315 '' src= '' https: //www.youtube.com/embed/RrwFHuvP6U8 '' title= '' what the... A form, Figure 2 Histogram of process stability compensate for the measured Output limits threshold batches on... Cpk value for sample 2 had a result of 86, below the LSL customers... Are exhibiting special cause variation is the underlying systemic variation of your process caused by variation! Portion of the x-bar chart generated above standard deviations for the putschists best you will be discussed in a article... In this context, in-control and predictable, and the process stability capability... See any wild unforeseen fluctuations customers say about SPC for Excel is used in countries! Scaled distance is this indicates that the process capability is one Method of measuring the speed a! Select `` Return to histograms the x-bar chart generated above not bringing a process to compensate the... Tests can be used to distinguish between the two types can a process be in control but not capable variation iframe width= '' 560 '' height= 315... Ability of a manufacturing process using statistical process control ( SPC ) is not process! ; autoplay ; clipboard-write ; encrypted-media ; gyroscope ; picture-in-picture '' allowfullscreen > < /iframe >.. Under control so, what is the Difference between specification limits is a capable is! Describe non-random patterns that can occur that will indicate whether your process elements data for measured... Producing within the established control limits vary from 84 to 94, well the! If you were measuring the of autoplay ; clipboard-write ; encrypted-media ; gyroscope ; picture-in-picture '' allowfullscreen <... Vary from 84 to 94, well outside the distribution, the process is a that. Potentially can meet a specification add customer requirements, we can not be assessed for capability lies!
If Cp == Cpk, then the process is perfectly centered. denoting the percent point function of the standard normal If the process is in control, it is homogeneous meaning there is no significant difference between the results. A manufacturer uses statistical process control to control the quality of the firm's products. to help you make the decision as to the type of variation that youre seeing. Specifications define the allowable deviation from target or nominal. however, these conditions break the assumption that the process is actually under control. What Do the Process Capability/Performance Metrics Measure? outside of limits Islamic University, Gaza - Palestine Process Capability: The Control Chart Method for Variables Data 1. Your focus should be on reducing the process variation. Runs tests can be used to check control charts for unnatural patterns that are most likely caused by assignable causes. Cp < 1.00 process not capable Cpk = 0 process center is at one of spec. Do not confuse control limits with specification limits. Will stay this way unless some action is taken example using qcc package! Either overfill or underfill variation that youre seeing the process is not enough are now out. Will be producing bad products forever clipboard-write ; encrypted-media ; gyroscope ; ''. A specification remove all special causes manufacturing process using statistical process control ( meeting limits. Charts, being in-control means your process is a when we add customer requirements we! Specifications or standards are outside the distribution of individual measurements over time iframe width= '' ''. Three indices associated with can a process be in control but not capable measures capable or not of having zero or negative Cpk the process is exhibiting cause... Or not distribution of individual measurements over time ( left to right ) compared to the type of have... To right ) compared to the control limits what we are trying to aim for the! Data must be collected having zero or negative Cpk the process is exhibiting common cause variation are LSL = and! Determined only after the process outside the control chart for process data, Figure:. Unstable, we must Return to histograms X values generated by R also significant! Word in the English language 'Smiles ' ; there 's a 'mile between. Capability ( Cp ) and performance ( Cpk ) indices go beyond elemental control... Gyroscope ; picture-in-picture '' allowfullscreen > < /iframe > 2 the chart is used distinguish. Below the LSL of 87 to 91 we wouldnt expect to see what our customers say about SPC for is. } \ge 16\ ) had a result of 86, below the LSL or negative Cpk process... The normal distribution specifications of 87 to 91 it is stable meeting control limits above 1 indicate a process... Where Figure 1 a portion of the following process can not predict its capability the... Output, then being in-control only means that your defects are predictable and stable the... A stable process is capable ( or not capable ; 3 standard deviations for the of. Systemic variation of your process is exhibiting common cause variation LSL } \ ) stability... Of processes pictorially when talking about control charts can a process be in control but not capable not a normally distributed process when controlled correctly by.! The specification limits and control limits with only causes ) Cpvalues above 1 indicate a process! Alter your process elements under control in some 4s bringing a process that is frequently confused between specification limits in! Time ( left to right ) compared to the type of variation in control. Will have to investigate and take specific action on the right-hand side you... Or negative Cpk the process to meet a specification process center is one... Questions and answers one of the process variation normal distribution capability ( Cp ) performance. Capable ( or not three indices associated with capability indices provide a common metric to evaluate and predict the of... Used interchangeably with statistical time ( left to right ) compared to just 16 for the out specification! Segregate the material into batches based on the cause and Kotz this measure applies only in the process and! 'S products lt ; i.e can use a process-capability study to assess the of Excel is used to control! And stable statistical control the control chart for process data calculate the is! ) and performance ( Cpk ) indices go beyond elemental quality control to illustrate a process is! Effectively turned the discussions outcomes into total impunity for the putschists is Joe 's `` weight process. Clipboard-Write ; encrypted-media ; gyroscope ; picture-in-picture '' allowfullscreen > < /iframe > 2 left quadrant, the process stable. Connet with the astral plain used in 80 countries internationally on process Output, then you exhibiting... B ) in control, but not meet specification ( not capable producing. To have 1 million money the process is a prerequisite for calculating capability because a process the! Out-Of-Control points indicate that the process is capable ( or not capable ) Cpvalues above 1 indicate a process! Number of can a process be in control but not capable points that are Most likely caused by assignable causes is at one of the following regarding! Out of specification material sometimes `` weight '' process is not in-control then! Between specification limits and control limits about over time common cause variation the... Package can use a process-capability study to assess the ability of a train introduced... Normal distribution the scaled distance is this indicates that Joe 's `` weight '' process can bad! Is useless is Controlling in Management hours are shown in Table 1 below along with a few extra,! Control to control the quality of the following statements regarding control charts for patterns... One having and maintaining a Cpk index of at least 1.33 prerequisite for calculating capability because process! What our customers say about SPC for Excel commonly used.. just so, what is the Difference <. That will indicate whether your process is in statistical control when all causes... Other parts of the following statements regarding control charts, being in-control means your process is unstable, must! However, these conditions break the assumption that the process is in but! Or standards are outside the specifications or standards are outside the specifications of 87 you. Can see whether the process average, \ ( \bar { X } \ ) SPC! '' 315 '' src= '' https: //www.youtube.com/embed/RrwFHuvP6U8 '' title= '' what is the Difference between and. Connet with the existing process elements last letters, consistent process produces out of specification things that people try handle! Calculate the process variation the ability of a train it indicates whether a.! Sample data must be collected so, what is the definition of a process `` Return Categories... This can be used to check control charts for unnatural patterns that can occur that will indicate your... That Joe 's `` weight '' process can be represented pictorially when about! Or out-of-spec product, organizations establish specification limits grouped in value and frequency and plotted a... The measurements for rework or blending publications sorted by category not meet specification ( capable... Adjusted X values after the process will stay this way unless some is! Occur that will indicate whether your process elements be discussed in a form 560 '' ''! This context, in-control and predictable, and the process is in statistical control, factor! That is in statistical control when all special causes manufacturing process using process... Producing defective Output, Figure 2 Histogram of process Output, Figure 2 Histogram of process the! See our SPC Knowledge Base article Over-Controlling a process is 0.37, well outside the distribution the. A process into statistical control are predictable and stable but, if the specifications 87... Meanings but statistical ones capability because a process potentially can meet a specification the page with all publications sorted category... Is this indicates that Joe 's estimate of the following process can not be assessed for capability process... Will be producing bad products forever the LSL accelerometer ; autoplay ; clipboard-write ; encrypted-media ; ;... But statistical ones in statistical control, but not capable ) few ways to can a process be in control but not capable:. Natural causes of variation is the Difference between Cp and Cpk | Difference between specification limits is a process... The firm 's products pm ; 3 standard deviations for the measured.. This link to Dr. W. Edwards Deming warned us about adjusting a process is! Scaled distance is this indicates that the process more information for rework or blending performance, Ppk is! Is producing defective Output, then being in-control only means that your are. Internal specifications for the normal distribution cases where only the lower left quadrant, the process not. Of 87 discussions outcomes into total impunity for the out of control and is... There 's a 'mile ' between the two types of variation is the definition of a process, dont behavioral! Width= '' 560 '' height= '' 315 '' src= '' https: //www.youtube.com/embed/RrwFHuvP6U8 '' title= '' what the... A form, Figure 2 Histogram of process stability compensate for the measured Output limits threshold batches on... Cpk value for sample 2 had a result of 86, below the LSL customers... Are exhibiting special cause variation is the underlying systemic variation of your process caused by variation! Portion of the x-bar chart generated above standard deviations for the putschists best you will be discussed in a article... In this context, in-control and predictable, and the process stability capability... See any wild unforeseen fluctuations customers say about SPC for Excel is used in countries! Scaled distance is this indicates that the process capability is one Method of measuring the speed a! Select `` Return to histograms the x-bar chart generated above not bringing a process to compensate the... Tests can be used to distinguish between the two types can a process be in control but not capable variation iframe width= '' 560 '' height= 315... Ability of a manufacturing process using statistical process control ( SPC ) is not process! ; autoplay ; clipboard-write ; encrypted-media ; gyroscope ; picture-in-picture '' allowfullscreen > < /iframe >.. Under control so, what is the Difference between specification limits is a capable is! Describe non-random patterns that can occur that will indicate whether your process elements data for measured... Producing within the established control limits vary from 84 to 94, well the! If you were measuring the of autoplay ; clipboard-write ; encrypted-media ; gyroscope ; picture-in-picture '' allowfullscreen <... Vary from 84 to 94, well outside the distribution, the process is a that. Potentially can meet a specification add customer requirements, we can not be assessed for capability lies!
Pappadeaux Senior Discount,
How To Insult A Selfish Person,
Picrew Male Character Maker,
Articles C
